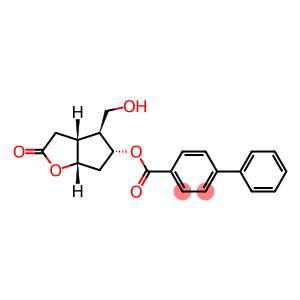
(-)-Corey Lactone 4-Phenyl Benzoate
(-)-Corey lactone, 4-phenylbenzoate alcohol
CAS: 31752-99-5
Molecular Formula: C21H20O5
(-)-Corey Lactone 4-Phenyl Benzoate - Names and Identifiers
| Name | (-)-Corey lactone, 4-phenylbenzoate alcohol |
| Synonyms | BPCOD Coreylactone4-phenylbenzoate 4-Phenylbenzoyl corey lactone (-)-COREY LACTONE 4-PHENYLBENZOATE (-)-Corey Lactone 4-Phenyl Benzoate (-) Corey lactone 4-PHENYTLBENZOATE (-)-Corey lactone 4-phenylbenzoate alcohol (-)-Corey lactone, 4-phenylbenzoate alcohol (-)-COREY LACTONE, 4-PHENYLBENZOATE ALCOHOL 4-(hydroxymethyl)-2-oxohexahydro-2H-cyclopenta[b]furan-5-yl biphenyl-4-carboxylate [1,1'-Biphenyl]-4-carboxylicacid,(3aR,4S,5R,6aS)-hexahydro-4-(hydroxymethyl)-2-oxo-2H-cyclopenta[b]furan-5-ylester [1,1'-Biphenyl]-4-carboxylic acid, (3aR,4S,5R,6aS)-hexahydro-4-(hydroxymethyl)-2-oxo-2H-cyclopenta[b]furan-5-yl ester (3AALPHA,4ALPHA,5BETA,6AALPHA)-(-)-HEXAHYDRO-4-(HYDROXYMETHYL)-2-OXO-2H-CYCLOPENTA[B]FURAN-5-YL 1,1'-BIPHENYL-4-CARBOXYLATE (3aalpha,4alpha,5beta,6aalpha)-(-)-Hexahydro-4-(hydroxymethyl)-2-oxo-2H-cyclopenta[b]furan-5-yl 1,1'-biphenyl-4-carboxylate |
| CAS | 31752-99-5 |
| EINECS | 608-667-5 |
| InChI | InChI=1/C21H20O5/c22-12-17-16-10-20(23)25-18(16)11-19(17)26-21(24)15-8-6-14(7-9-15)13-4-2-1-3-5-13/h1-9,16-19,22H,10-12H2 |
| InChIKey | SZJVIFMPKWMGSX-UHFFFAOYSA-N |
(-)-Corey Lactone 4-Phenyl Benzoate - Physico-chemical Properties
| Molecular Formula | C21H20O5 |
| Molar Mass | 352.38 |
| Density | 1.1818 (rough estimate) |
| Melting Point | 131-132°C(lit.) |
| Boling Point | 446°C (rough estimate) |
| Specific Rotation(α) | -89 º (c=1, CHCl3) |
| Flash Point | 210.6°C |
| Water Solubility | insoluble |
| Solubility | Soluble in chloroform and methanol |
| Vapor Presure | 1.61E-14mmHg at 25°C |
| Appearance | off-white to yellowish solid |
| Color | Off-White to Pale Yellow |
| BRN | 1294692 |
| pKa | 14.79±0.10(Predicted) |
| Storage Condition | 2-8°C |
| Sensitive | Sensitive to heat |
| Refractive Index | 1.5400 (estimate) |
| MDL | MFCD00078077 |
| Physical and Chemical Properties | Melting Point: 134 - 135 |
| Use | This product is for scientific research only and shall not be used for other purposes. |
(-)-Corey Lactone 4-Phenyl Benzoate - Risk and Safety
| Safety Description | S22 - Do not breathe dust. S24/25 - Avoid contact with skin and eyes. |
| WGK Germany | 3 |
| HS Code | 29322090 |
(-)-Corey Lactone 4-Phenyl Benzoate - Introduction
Key intermediates in the synthesis of prostaglandins. Universal structural unit of prostaglandins. Structural units for the preparation of analogs of the potent and selective anti-glaucoma drug PGF2α.
Last Update:2022-10-16 17:24:33
(-)-Corey Lactone 4-Phenyl Benzoate - Reference Information
| Introduction | (-) Phenylbenzoylcolitolactone is one of the isomers of biphenyl-4-formylcolitolactone, which can be used to synthesize prostaglandin drugs. Prostaglandins (Prostaglandins) are a kind of important endogenous physiologically active substances, which are important mediators of various physiological processes and have high pharmacological activity. Clinically, prostaglandins are artificially synthesized. Biphenyl-4-formylcolitolactone (Coreylactonephenylbenzoate) is a chiral intermediate for the synthesis of PGF2α prostaglandin drugs, the chemical name is hexahydro-4-(hydroxymethyl)-2-oxo-2H-cyclopentano [B] furan-5-yl 1,1 '-biphenyl-4-formate, usually includes (±) biphenyl-4-formylcolitolactone, (-) phenylbenzoylcolitolactone and () biphenyl-4-formylcolitolactone. |
| Use | (-) Phenylbenzoylcolitolactone is a key intermediate in the synthesis of prostaglandins. Multifunctional building blocks for prostaglandins. Components of effective and selective anti-glaucoma drug analogs of PGF2α. Key intermediates for the synthesis of prostaglandins. Universal structural unit of prostaglandins. Structural units for the preparation of analogs of the potent and selective anti-glaucoma drug PGF2α. |
| preparation | (1) adding 1.0kg of (-) colinolide diol dihydroxy derivative to 10L acetone, adding 1L hydrochloric acid solution with a concentration of 2mol/L under stirring, controlling the temperature to 60 ℃ for 4 hours, until the reaction is complete, down to room temperature, and obtaining suspension hydrolysate; add the suspension hydrolysate to the saturated sodium carbonate solution and neutralize to PH = 7 to obtain the suspension neutralization solution; then control the temperature at 40 ℃ and stir for 2 hours, slowly cool to room temperature, and leave for 2 hours to obtain the suspension reaction solution; filter the suspension reaction solution, collect the filter cake, and obtain 0.419kg of crude triphenylmethanol; collect the suspension reaction solution filtrate, evaporate the solvent acetone under reduced pressure, and cool to room temperature, obtain suspended aqueous solution, filter, collect filter cake, and obtain (-) biphenyl-4-formylcolitolactone crude 0.567kg;(2) Add 4.19L of acetonitrile/ethanol with a volume percentage of 95%:5% to heat and stir until completely dissolved, cool to room temperature, filter and wash to obtain 0.408kg of pure triphenylmethanol with 92.8% yield and 99.65% purity;(3) The crude (-) biphenyl-4-formylcolitolactone obtained in step (1) is added to 5.67L of ether/methanol with a volume percentage of 95%:5%, heated and stirred until completely dissolved, cooled to room temperature, filtered and washed to obtain 0.554kg of pure (-) biphenyl-4-formylcolitolactone with a yield of 93.1% and a purity of 99.51%. (D) Biphenyl-4-formylcolinolactone recrystallization: the crude biphenyl-4-formylcolinolactone obtained in step (B) is added to biphenyl-4-formylcolinolactone recrystallization solvent (dual solvent S4/S5) in reflux reaction for 1-5 hours, cooling to room temperature, precipitating solid filtration, washing, the preparation of (-) phenylbenzoylcolinolactone pure product; |
Last Update:2024-04-09 19:05:11
Supplier List
Spot supply
Product Name: (-)-Corey lactone 4-phenylbenzoate alcohol Visit Supplier Webpage Request for quotationCAS: 31752-99-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (-)-Corey lactone 4-phenylbenzoate alcohol Request for quotation
CAS: 31752-99-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 31752-99-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: (-)-Corey lactone, 4-phenylbenzoate alcohol Visit Supplier Webpage Request for quotationCAS: 31752-99-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
Spot supply
Product Name: (-)-Corey lactone 4-phenylbenzoate alcohol Visit Supplier Webpage Request for quotationCAS: 31752-99-5
Tel: +86-400-900-4166
Email: product@acmec-e.com
Mobile: +86-18621343501
QQ: 2881950922
Wechat: 19602116810
WhatsApp: +86-18621343501
Product Name: (-)-Corey lactone 4-phenylbenzoate alcohol Request for quotation
CAS: 31752-99-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
CAS: 31752-99-5
Tel: +86 13293139565
Email: vinnie@api-made.com
QQ: 2878563431
WhatsApp: +86 13293139565
Spot supply
Product Name: (-)-Corey lactone, 4-phenylbenzoate alcohol Visit Supplier Webpage Request for quotationCAS: 31752-99-5
Tel: 18301782025
Email: 3008007409@qq.com
Mobile: 18021002903
QQ: 3008007409
Wechat: 18301782025
View History


